
Drug Monitoring
medMATCH
®
Report

Should be easy to implement and based on
clear, actionable insights.
Our medMATCH
®
report is designed to provide actionable insights with the goal of
“at-a-glance” interpretation.
Read on to learn more about the Drug Monitoring medMATCH
®
report.
• Inspired by your feedback, designed to meet your needs
• Simple and intuitive
• Allows for fast and easy interpretation of results
Critical care decisions

Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Patient ID: 0131627N
Specimen: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Test Ordered Result Cutoff medMATCH Lab
Gabapentin
NEGATIVE 1000 ng/mL
INCONSISTENT
Gabapentin Comments: See LDT Notes
Test Ordered Result Cutoff Lab
DRUG MONITOR, FENTANYL, SCREEN, URINE IG
Fentanyl NEGATIVE
See Note 1
0.5 ng/mL
Test Ordered Result Reference Range Lab
SPECIMEN VALIDITY TEST IG
Creatinine 26.7 > or = 20.0 mg/dL
pH 7.8 4.5-9.0
Oxidant NEGATIVE <200 mcg/mL
NOTES AND COMMENTS
This drug testing is for medical treatment only. Analysis was performed as non-forensic testing and these results should be used only by healthcare providers to
render diagnosis or treatment, or to monitor progress of medical conditions.
Note 1:
The results are presumptive; based only on screening methods, and they have not been confirmed by a definitive method.
Alcohol Metab Notes:
Ethyl sulfate detected is consistent with exposure to alcohol.
Buprenorphine Notes:
Buprenorphine, Norbuprenorphine detected is consistent with the use of the drug(s) Buprenorphine or Buprenorphine with Naloxone. Naloxone may be negative
due to poor oral bioavailability and/or short half-life.
Buprenorphine, Norbuprenorphine, Naloxone detected is consistent with the use of the drug Buprenorphine with Naloxone.
LDT Notes:
Confirmation tests were developed and their analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or
approved by the FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
medMATCH(R) enables providers to identify if drug use is consistent or inconsistent with a corresponding prescribed medication(s) list.
Healthcare Providers needing Interpretation assistance, please contact us at 1.877.40.RXTOX (1.877.407.9869) M-F, 8am to 10pm EST
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
* TEST CLIENT (HQ) DAL has requested a copy of this report be sent to you. Ordering Physician: COLMENAR, ANTONIO
PAGE 2 OF 2
Report Status: Final
BROCHURE, REPORT2
Patient Information Specimen Information Client Information
BROCHURE, REPORT2
DOB: Not Given AGE: 37
Gender: M Fasting: Y
Patient ID: 0131630N
Specimen: IF048805B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:59 EDT
Reported: 09/29/2020 / 13:13 EDT
(* A Copy From)
Client #: 97502882
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048805B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Test Ordered Result Cutoff medMATCH Lab
Morphine NEGATIVE 50 ng/mL
Norhydrocodone 1273 50 ng/mL
CONSISTENT
Opiates Comments: See Opiates Notes, LDT Notes
Oxycodone NEGATIVE 100 ng/mL
Phencyclidine NEGATIVE 25 ng/mL
DRUG MONITOR, TRICYCLIC ANTIDEPRESS, QN, URINE IG
Amitriptyline
200 100 ng/mL
CONSISTENT
Nortriptyline 300 100 ng/mL
CONSISTENT
Tricyclic Antidep Comment: See Tricyclic Antidep Notes, LDT Notes
Test Ordered Result Reference Range Lab
SPECIMEN VALIDITY TEST IG
Creatinine 26.6 > or = 20.0 mg/dL
pH 4.5 4.5-9.0
Oxidant POSITIVE A <200 mcg/mL
NOTES AND COMMENTS
This drug testing is for medical treatment only. Analysis was performed as non-forensic testing and these results should be used only by healthcare providers to
render diagnosis or treatment, or to monitor progress of medical conditions.
Benzodiazepines Notes:
aOH Alprazolam detected is consistent with the use of the drug Alprazolam.
Temazepam detected is consistent with the use of the drug Temazepam. Temazepam can be a prescribed drug and is also a metabolite of Diazepam. The
metabolite Oxazepam is not present at or above the cutoff.
Temazepam detected is consistent with the use of the drug Temazepam. Temazepam can be a prescribed drug and is also a metabolite of Diazepam.
Opiates Notes:
Hydrocodone, Norhydrocodone detected is consistent with the use of the drug Hydrocodone. The metabolite Hydromorphone is not present at or above the cutoff.
Tricyclic Antidep Notes:
Amitriptyline, Nortriptyline detected is consistent with the use of the drug Amitriptyline.
Nortriptyline detected is consistent with the use of the drug Nortriptyline. Nortriptyline can be a prescribed drug and is also a metabolite of Amitriptyline.
LDT Notes:
Confirmation tests were developed and their analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or
approved by the FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
medMATCH(R) enables providers to identify if drug use is consistent or inconsistent with a corresponding prescribed medication(s) list.
Healthcare Providers needing Interpretation assistance, please contact us at 1.877.40.RXTOX (1.877.407.9869) M-F, 8am to 10pm EST
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
PAGE 2 OF 2
Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
MedMATCH
®
Drug Monitoring Report
Click on any highlighted section to learn more
about the new report features.
+
+
+
Sample report:

Report Status: Final
REPORT3
Patient Information Specimen Information Client Information
SAMPLE REPORT3
DOB: Not Given AGE: 40
Gender: M Fasting: Y
Phone: NG
Patient ID: 0131634N
Specimen: IF048807B
Requisition: 0131634
Lab Ref #: IF048807B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 13:10 EDT
Reported: 09/29/2020 / 13:19 EDT
Client #: 97502882
TEST
Attn: Dr. Jones
Main Street
City, NJ 07000
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048807B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Drug Monitoring Report
Test Ordered Result Cutoff Lab
DRUG MONITOR,AMPHETAMINE, W/CONF, W/DL,URINE IG
Amphetamines POSITIVE 500 ng/mL
Amphetamine 300 250 ng/mL
Methamphetamine 500 250 ng/mL
Amphetamines Comments: See Amphetamines Notes, LDT Notes
DRUG MONITOR, METHAMPHETAMINE D/L,U IG
d Methamphetamine 80 %
l Methamphetamine 20 %
D/L Methamphet Comments: See D/L Methamphet Notes, LDT Notes
NOTES AND COMMENTS
This drug testing is for medical treatment only. Analysis was performed as non-forensic testing and these results should be used only by healthcare providers to
render diagnosis or treatment, or to monitor progress of medical conditions.
Amphetamines Notes:
Amphetamine detected is consistent with the use of the drug Amphetamine. Amphetamine can be a prescribed drug and is also a metabolite of methamphetamine.
Amphetamine, Methamphetamine detected is consistent with the use of the drug Methamphetamine.
D/L Methamphet Notes:
d-Methamphetamine > 20% detected is consistent with the use of the drug(s) prescription Methamphetamine, Benzphetamine or Methamphetamine from a
clandestine source.
LDT Notes:
Confirmation tests were developed and their analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or
approved by the FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
Healthcare Providers needing Interpretation assistance, please contact us at 1.877.40.RXTOX (1.877.407.9869) M-F, 8am to 10pm EST
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
PAGE 1 OF 1
Drug Monitoring Report
Click on any highlighted section to learn more
about the new report features.
+
Sample report:

Results summarized
Using the summary table, you can quickly find and address key
elements of your patient’s report results.
• The medMATCH
®
Summary provides quick, at-a-glance results in a table at the top of the report.
• Results are categorized based on the combination of clinician-provided prescription data and test results.
Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
+
+
+

Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
Result details
The body of the medMATCH
®
report provides the individual test results,
with key results identified with icons and color to aid in fast and intuitive
result interpretation.
• Next to a drug indicates that it was a prescribed drug
• Blue test name and a indicates a prescribed drug with a consistent result
• Red test name and an indicates a prescribed drug with an inconsistent result
+
+
+
+

Report Status: Final
BROCHURE, REPORT2
Patient Information Specimen Information Client Information
BROCHURE, REPORT2
DOB: Not Given AGE: 37
Gender: M Fasting: Y
Patient ID: 0131630N
Specimen: IF048805B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:59 EDT
Reported: 09/29/2020 / 13:13 EDT
(* A Copy From)
Client #: 97502882
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048805B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Test Ordered Result Cutoff medMATCH Lab
Morphine NEGATIVE 50 ng/mL
Norhydrocodone 1273 50 ng/mL
CONSISTENT
Opiates Comments: See Opiates Notes, LDT Notes
Oxycodone NEGATIVE 100 ng/mL
Phencyclidine NEGATIVE 25 ng/mL
DRUG MONITOR, TRICYCLIC ANTIDEPRESS, QN, URINE IG
Amitriptyline
200 100 ng/mL
CONSISTENT
Nortriptyline 300 100 ng/mL
CONSISTENT
Tricyclic Antidep Comment: See Tricyclic Antidep Notes, LDT Notes
Test Ordered Result Reference Range Lab
SPECIMEN VALIDITY TEST IG
Creatinine 26.6 > or = 20.0 mg/dL
pH 4.5 4.5-9.0
Oxidant POSITIVE A <200 mcg/mL
NOTES AND COMMENTS
This drug testing is for medical treatment only. Analysis was performed as non-forensic testing and these results should be used only by healthcare providers to
render diagnosis or treatment, or to monitor progress of medical conditions.
Benzodiazepines Notes:
aOH Alprazolam detected is consistent with the use of the drug Alprazolam.
Temazepam detected is consistent with the use of the drug Temazepam. Temazepam can be a prescribed drug and is also a metabolite of Diazepam. The
metabolite Oxazepam is not present at or above the cutoff.
Temazepam detected is consistent with the use of the drug Temazepam. Temazepam can be a prescribed drug and is also a metabolite of Diazepam.
Opiates Notes:
Hydrocodone, Norhydrocodone detected is consistent with the use of the drug Hydrocodone. The metabolite Hydromorphone is not present at or above the cutoff.
Tricyclic Antidep Notes:
Amitriptyline, Nortriptyline detected is consistent with the use of the drug Amitriptyline.
Nortriptyline detected is consistent with the use of the drug Nortriptyline. Nortriptyline can be a prescribed drug and is also a metabolite of Amitriptyline.
LDT Notes:
Confirmation tests were developed and their analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or
approved by the FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
medMATCH(R) enables providers to identify if drug use is consistent or inconsistent with a corresponding prescribed medication(s) list.
Healthcare Providers needing Interpretation assistance, please contact us at 1.877.40.RXTOX (1.877.407.9869) M-F, 8am to 10pm EST
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
PAGE 2 OF 2
Result details
Contextual information is consolidated to make individual
recommendations and test results readily accessible.
+
+

Results history
Data includes results meeting strict identification matching.
Our results history feature displays results for the current order as well as
results from the prior 4 orders, thus providing additional insights and allowing you to easily
compare current results with historical results and further aid patient care decisions.
Patient Information Specimen Information Client Information
Fasting: Y
Specimen:
Collected: 09/24/2020 / 12:00 CDT
Received: 09/24/2020 / 19:17 CDT
Reported: 09/25/2020 / 03:58 CDT
CLIENT SERVICES: 866.697.8378
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
medMATCH
= Consistent = Inconsistent
= Prescribed
Test Name
09/24/2020
09/22/2020 09/19/2020 09/17/2020
Marijuana Metabolite POSITIVE POSITIVE NEGATIVE CONFIRMED POSITIVE
Marijuana Metabolite 5
5 NEGATIVE 100
Methadone Metabolite POSITIVE POSITIVE POSITIVE POSITIVE
EDDP 100 NEGATIVE
100 100
Methadone 100
100
NEGATIVE
100
Oxycodone POSITIVE POSITIVE
NEGATIVE
POSITIVE
Noroxycodone 100
100
-
100
Oxycodone 100
100
-
100
Oxymorphone 100
100
-
100
Specimen Validity Testing
Specific Gravity 1.002 L 1.002 L 1.002 L 1.002 L
pH 6.7 6.7 6.7 6.7
Creatinine 11.2 L 12.3 L 14.5 L 11.2 L
Oxidant NEGATIVE NEGATIVE NEGATIVE NEGATIVE
Only results from the current report and the past 4 orders with Quest Diagnostics are displayed (Data includes results meeting strict identification matching). Table displays presumptive
results first then corresponding definitive results. Dash indicates a presumptive test was negative therefore definitive testing was not needed, or the test was not ordered by the clinician
in subsequent testing.
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
LIST OF RESULTS PRINTED IN THE OUT OF RANGE COLUMN:
Creatinine 11.2 L
> or = 20.0 mg/dL IG
Specific Gravity 1.002 L
> or = 1.003 IG
PAGE 3 OF 3
+
+
+
+
+

Report Status: Final
REPORT3
Patient Information Specimen Information Client Information
SAMPLE REPORT3
DOB: Not Given AGE: 40
Gender: M Fasting: Y
Phone: NG
Patient ID: 0131634N
Specimen: IF048807B
Requisition: 0131634
Lab Ref #: IF048807B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 13:10 EDT
Reported: 09/29/2020 / 13:19 EDT
Client #: 97502882
TEST
Attn: Dr. Jones
Main Street
City, NJ 07000
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048807B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Drug Monitoring Report
Test Ordered Result Cutoff Lab
DRUG MONITOR,AMPHETAMINE, W/CONF, W/DL,URINE IG
Amphetamines POSITIVE 500 ng/mL
Amphetamine 300 250 ng/mL
Methamphetamine 500 250 ng/mL
Amphetamines Comments: See Amphetamines Notes, LDT Notes
DRUG MONITOR, METHAMPHETAMINE D/L,U IG
d Methamphetamine 80 %
l Methamphetamine 20 %
D/L Methamphet Comments: See D/L Methamphet Notes, LDT Notes
NOTES AND COMMENTS
This drug testing is for medical treatment only. Analysis was performed as non-forensic testing and these results should be used only by healthcare providers to
render diagnosis or treatment, or to monitor progress of medical conditions.
Amphetamines Notes:
Amphetamine detected is consistent with the use of the drug Amphetamine. Amphetamine can be a prescribed drug and is also a metabolite of methamphetamine.
Amphetamine, Methamphetamine detected is consistent with the use of the drug Methamphetamine.
D/L Methamphet Notes:
d-Methamphetamine > 20% detected is consistent with the use of the drug(s) prescription Methamphetamine, Benzphetamine or Methamphetamine from a
clandestine source.
LDT Notes:
Confirmation tests were developed and their analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or
approved by the FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
Healthcare Providers needing Interpretation assistance, please contact us at 1.877.40.RXTOX (1.877.407.9869) M-F, 8am to 10pm EST
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
PAGE 1 OF 1
Result details
Drug Monitoring Report
When results are ordered without medMATCH, the Drug Monitoring report will be issued.
The body of the Drug Monitoring report provides the individual test results, with key
results identified with icons and color to aid in fast and intuitive result interpretation.
Red test name and result indicate a positive test result.
+
+

See the difference Quest can make.
Our state-of-the-art offering begins with a comprehensive menu and features exceptional service and
reporting, as well as expert support through our dedicated Rx Tox line at 1.877.40.RXTOX (1.877.407.9869)
As an industry leader, Quest is committed to continuous improvement and to providing you with the best drug
monitoring experience possible.
Visit QuestDrugMonitoring.com or talk to your sales representative at 1.866.MYQUEST (1.866.697.8378)
We’re ready to assist you
Image content features models and is intended for illustrative purposes only.
QuestDiagnostics.com
Quest, Quest Diagnostics, any associated logos, and all associated Quest Diagnostics registered or unregistered trademarks
are the property of Quest Diagnostics. All third-party marks - ® and ™ - are the property of their respective owners.
© 2021 Quest Diagnostics Incorporated. All rights reserved.
MI10063 2/2021.

Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
+
+
+
Results summarized
• The medMATCH® Summary provides quick, at-a-glance results in a table at the top of the report.
• Results are categorized based on the combination of clinician provided prescription data and test results
X
Prescribed drugs
The prescribed drugs section has been divided into 2 categories:
• Prescribed Consistent: the prescribed drugs are detected; the results are
consistent with treatment.
• Prescribed Inconsistent: the prescribed drugs are not detected; the
results are inconsistent with treatment.
Using the summary table, you can quickly find and address key
elements of your patient’s report results.

Results summarized
• The medMATCH
®
Summary provides quick, at-a-glance results in a table at the top of the report.
• Results are categorized based on the combination of clinician provided prescription data and test results
Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
+
+
+
X
Not prescribed, inconsistent
• Positive drug results for non-prescribed drugs or substances are listed.
This alerts the clinician to potential drug misuse.
• In this example, non-prescribed drugs or substances were detected; the
results are inconsistent with treatment.
Using the summary table, you can quickly find and address key
elements of your patient’s report results.

Using the summary table, you can quickly find and address key
elements of your patient’s report results.
Results summarized
• The medMATCH
®
Summary provides quick, at-a-glance results in a table at the top of the report.
• Results are categorized based on the combination of clinician provided prescription data and test results
Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
+
+
+
X
Prescriptions without testing
Test alert is triggered if a prescription drug is provided, but a test was
not ordered.
This serves as an alert to the clinician to non-tested prescription drugs.

Result details
The body of the medMATCH
®
report provides the individual test results,
with key results identified with icons and color to aid in fast and intuitive
result interpretation.
Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
• Next to a drug indicates that it was a prescribed drug
• Blue test name and a indicates a prescribed drug with a consistent result
• Red test name and an indicates a prescribed drug with an inconsistent result
+
+
+
+
X
Drug Class notes
Contextual information is consolidated and displayed at the end of the report,
to make individual recommendations and test results readily accessible.
(Notes and Comments section)
Example for alcohol metabolites:
“Ethyl sulfate detected is consistent with exposure to alcohol.”

Result details
The body of the medMATCH
®
report provides the individual test results,
with key results identified with icons and color to aid in fast and intuitive
result interpretation.
Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
• Next to a drug indicates that it was a prescribed drug
• Blue test name and a indicates a prescribed drug with a consistent result
• Red test name and an indicates a prescribed drug with an inconsistent result
+
+
+
+
X
Inconsistent flag
When medMATCH
®
is ordered and results do not match prescribed
medications, the inconsistent flag is triggered.

Result details
The body of the medMATCH
®
report provides the individual test results,
with key results identified with icons and color to aid in fast and intuitive
result interpretation.
Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
• Next to a drug indicates that it was a prescribed drug
• Blue test name and a indicates a prescribed drug with a consistent result
• Red test name and an indicates a prescribed drug with an inconsistent result
+
+
+
+
X
LDT notes
Laboratory-developed test, or LDT, notes are consolidated into a single note
at the end of the report. (Notes and Comments section)
Example Note
“Confirmation tests were developed and their analytical performance
characteristics have been determined by Quest Diagnostics. It has not been
cleared or approved by the FDA. This assay has been validated pursuant to
the CLIA regulations and is used for clinical purposes.”

Result details
The body of the medMATCH
®
report provides the individual test results,
with key results identified with icons and color to aid in fast and intuitive
result interpretation.
Report Status: Final
BROCHURE, REPORT
Patient Information Specimen Information Client Information
BROCHURE, REPORT
DOB: Not Given AGE: 40
Gender: F Fasting: Y
Phone: 123.432.1244
Patient ID: 0131627N
Specimen: IF048802B
Requisition: 0131627
Lab Ref #: IF048802B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:37 EDT
Reported: 09/29/2020 / 11:00 EDT
(* A Copy From)
Client #: 97502882
AARRSSS
TEST CLIENT (HQ) 5
Attn: ATTN: TEST DEPARTMENT
224 E 96TH ST
NEW YORK, NY 10128-3802
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048802B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Room: 12 Bed: 15
COMMENTS:
TESTING REPORT FOR BROCHURE
medMATCH® Drug Monitoring Report
Legend
medMATCH Summary
medMATCH indicates presence or absence of prescribed and non prescribed drugs. Prescribed drugs are provided by the ordering clinician.
= Consistent
Prescribed Consistent
Drugs prescribed and detected
Prescribed Inconsistent
Drugs prescribed but not detected
Not Prescribed Inconsistent
Drugs detected but not prescribed
= Inconsistent
= Prescribed
= Buprenorphine Nalox = Gabapentin = Ethyl Sulfate (ETS)
Prescriptions reported by client with no testing ordered: Zolpidem
Test Ordered Result Cutoff medMATCH Lab
DRUG MONITORING, PANEL 8 WITH CONFIRMATION, URINE IG
Alcohol Metabolites POSITIVE 500 ng/mL
Ethyl Glucuronide (ETG) NEGATIVE 500 ng/mL
Ethyl Sulfate (ETS) 100 100 ng/mL
INCONSISTENT
Alcohol Metab Comments: See Alcohol Metab Notes, LDT Notes
Amphetamines NEGATIVE 500 ng/mL
Benzodiazepines NEGATIVE 100 ng/mL
Buprenorphine POSITIVE 5 ng/mL
Buprenorphine
101 2 ng/mL
CONSISTENT
Norbuprenorphine
105 2 ng/mL
CONSISTENT
Naloxone
110 2 ng/mL
CONSISTENT
Buprenorphine Comments: See Buprenorphine Notes, LDT Notes
Cocaine Metabolite NEGATIVE CONFIRMED 150 ng/mL
Benzoylecgonine NEGATIVE 100 ng/mL
Cocaine Comments: See LDT Notes
6 Acetylmorphine NEGATIVE 10 ng/mL
Marijuana Metabolite NEGATIVE 20 ng/mL
Naloxone
110 2 ng/mL
CONSISTENT
MDMA NEGATIVE 500 ng/mL
Opiates NEGATIVE 100 ng/mL
Oxycodone NEGATIVE 100 ng/mL
DRUG MONITOR, GABAPENTIN, QN, URINE IG
PAGE 1 OF 2
• Next to a drug indicates that it was a prescribed drug
• Blue test name and a indicates a prescribed drug with a consistent result
• Red test name and an indicates a prescribed drug with an inconsistent result
+
+
+
+
X
Consistent flag
When medMATCH
®
is ordered and the prescribed drug is detected, the
consistent flag is triggered.

Report Status: Final
BROCHURE, REPORT2
Patient Information Specimen Information Client Information
BROCHURE, REPORT2
DOB: Not Given AGE: 37
Gender: M Fasting: Y
Patient ID: 0131630N
Specimen: IF048805B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:59 EDT
Reported: 09/29/2020 / 13:13 EDT
(* A Copy From)
Client #: 97502882
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048805B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Test Ordered Result Cutoff medMATCH Lab
Morphine NEGATIVE 50 ng/mL
Norhydrocodone 1273 50 ng/mL
CONSISTENT
Opiates Comments: See Opiates Notes, LDT Notes
Oxycodone NEGATIVE 100 ng/mL
Phencyclidine NEGATIVE 25 ng/mL
DRUG MONITOR, TRICYCLIC ANTIDEPRESS, QN, URINE IG
Amitriptyline
200 100 ng/mL
CONSISTENT
Nortriptyline 300 100 ng/mL
CONSISTENT
Tricyclic Antidep Comment: See Tricyclic Antidep Notes, LDT Notes
Test Ordered Result Reference Range Lab
SPECIMEN VALIDITY TEST IG
Creatinine 26.6 > or = 20.0 mg/dL
pH 4.5 4.5-9.0
Oxidant POSITIVE A <200 mcg/mL
NOTES AND COMMENTS
This drug testing is for medical treatment only. Analysis was performed as non-forensic testing and these results should be used only by healthcare providers to
render diagnosis or treatment, or to monitor progress of medical conditions.
Benzodiazepines Notes:
aOH Alprazolam detected is consistent with the use of the drug Alprazolam.
Temazepam detected is consistent with the use of the drug Temazepam. Temazepam can be a prescribed drug and is also a metabolite of Diazepam. The
metabolite Oxazepam is not present at or above the cutoff.
Temazepam detected is consistent with the use of the drug Temazepam. Temazepam can be a prescribed drug and is also a metabolite of Diazepam.
Opiates Notes:
Hydrocodone, Norhydrocodone detected is consistent with the use of the drug Hydrocodone. The metabolite Hydromorphone is not present at or above the cutoff.
Tricyclic Antidep Notes:
Amitriptyline, Nortriptyline detected is consistent with the use of the drug Amitriptyline.
Nortriptyline detected is consistent with the use of the drug Nortriptyline. Nortriptyline can be a prescribed drug and is also a metabolite of Amitriptyline.
LDT Notes:
Confirmation tests were developed and their analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or
approved by the FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
medMATCH(R) enables providers to identify if drug use is consistent or inconsistent with a corresponding prescribed medication(s) list.
Healthcare Providers needing Interpretation assistance, please contact us at 1.877.40.RXTOX (1.877.407.9869) M-F, 8am to 10pm EST
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
PAGE 2 OF 2
+
+
Result details
Contextual information is consolidated to make individual
recommendations and test results readily accessible.
X
Notes and Comments
Drug class and LDT notes are consolidated at the end of the report in the
Notes and Comments section.

Report Status: Final
BROCHURE, REPORT2
Patient Information Specimen Information Client Information
BROCHURE, REPORT2
DOB: Not Given AGE: 37
Gender: M Fasting: Y
Patient ID: 0131630N
Specimen: IF048805B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 10:59 EDT
Reported: 09/29/2020 / 13:13 EDT
(* A Copy From)
Client #: 97502882
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048805B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Test Ordered Result Cutoff medMATCH Lab
Morphine NEGATIVE 50 ng/mL
Norhydrocodone 1273 50 ng/mL
CONSISTENT
Opiates Comments: See Opiates Notes, LDT Notes
Oxycodone NEGATIVE 100 ng/mL
Phencyclidine NEGATIVE 25 ng/mL
DRUG MONITOR, TRICYCLIC ANTIDEPRESS, QN, URINE IG
Amitriptyline
200 100 ng/mL
CONSISTENT
Nortriptyline 300 100 ng/mL
CONSISTENT
Tricyclic Antidep Comment: See Tricyclic Antidep Notes, LDT Notes
Test Ordered Result Reference Range Lab
SPECIMEN VALIDITY TEST IG
Creatinine 26.6 > or = 20.0 mg/dL
pH 4.5 4.5-9.0
Oxidant POSITIVE A <200 mcg/mL
NOTES AND COMMENTS
This drug testing is for medical treatment only. Analysis was performed as non-forensic testing and these results should be used only by healthcare providers to
render diagnosis or treatment, or to monitor progress of medical conditions.
Benzodiazepines Notes:
aOH Alprazolam detected is consistent with the use of the drug Alprazolam.
Temazepam detected is consistent with the use of the drug Temazepam. Temazepam can be a prescribed drug and is also a metabolite of Diazepam. The
metabolite Oxazepam is not present at or above the cutoff.
Temazepam detected is consistent with the use of the drug Temazepam. Temazepam can be a prescribed drug and is also a metabolite of Diazepam.
Opiates Notes:
Hydrocodone, Norhydrocodone detected is consistent with the use of the drug Hydrocodone. The metabolite Hydromorphone is not present at or above the cutoff.
Tricyclic Antidep Notes:
Amitriptyline, Nortriptyline detected is consistent with the use of the drug Amitriptyline.
Nortriptyline detected is consistent with the use of the drug Nortriptyline. Nortriptyline can be a prescribed drug and is also a metabolite of Amitriptyline.
LDT Notes:
Confirmation tests were developed and their analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or
approved by the FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
medMATCH(R) enables providers to identify if drug use is consistent or inconsistent with a corresponding prescribed medication(s) list.
Healthcare Providers needing Interpretation assistance, please contact us at 1.877.40.RXTOX (1.877.407.9869) M-F, 8am to 10pm EST
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
PAGE 2 OF 2
+
+
Result details
Contextual information is consolidated to make individual
recommendations and test results readily accessible.
+
X
Additional assistance is available
The number for our dedicated, toxicology support line is indicated at the
bottom of the report. If you have a question or would like to consult with
our medical staff, clinicians can contact us using this number.

Results history
Data includes results meeting strict identification matching.
Patient Information Specimen Information Client Information
Fasting: Y
Specimen:
Collected: 09/24/2020 / 12:00 CDT
Received: 09/24/2020 / 19:17 CDT
Reported: 09/25/2020 / 03:58 CDT
CLIENT SERVICES: 866.697.8378
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
medMATCH
= Consistent = Inconsistent
= Prescribed
Test Name
09/24/2020
09/22/2020 09/19/2020 09/17/2020
Marijuana Metabolite POSITIVE POSITIVE NEGATIVE CONFIRMED POSITIVE
Marijuana Metabolite 5
5 NEGATIVE 100
Methadone Metabolite POSITIVE POSITIVE POSITIVE POSITIVE
EDDP 100 NEGATIVE
100 100
Methadone 100
100
NEGATIVE
100
Oxycodone POSITIVE POSITIVE
NEGATIVE
POSITIVE
Noroxycodone 100
100
-
100
Oxycodone 100
100
-
100
Oxymorphone 100
100
-
100
Specimen Validity Testing
Specific Gravity 1.002 L 1.002 L 1.002 L 1.002 L
pH 6.7 6.7 6.7 6.7
Creatinine 11.2 L 12.3 L 14.5 L 11.2 L
Oxidant NEGATIVE NEGATIVE NEGATIVE NEGATIVE
Only results from the current report and the past 4 orders with Quest Diagnostics are displayed (Data includes results meeting strict identification matching). Table displays presumptive
results first then corresponding definitive results. Dash indicates a presumptive test was negative therefore definitive testing was not needed, or the test was not ordered by the clinician
in subsequent testing.
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
LIST OF RESULTS PRINTED IN THE OUT OF RANGE COLUMN:
Creatinine 11.2 L
> or = 20.0 mg/dL IG
Specific Gravity 1.002 L
> or = 1.003 IG
PAGE 3 OF 3
+
+
+
+
+
X
Patient history
• Results from the current report and the past 4 orders (maximum) are displayed.
• To save space, only those drugs which have at least one positive result are
displayed.
• SVT (specimen validity test) results are always displayed if performed.
• Data includes results meeting strict identification matching.
Our results history feature displays results for the current order as well as
results from the prior 4 orders, thus providing additional insights and allowing you to easily
compare current results with historical results and further aid patient care decisions.

Results history
Data includes results meeting strict identification matching.
Patient Information Specimen Information Client Information
Fasting: Y
Specimen:
Collected: 09/24/2020 / 12:00 CDT
Received: 09/24/2020 / 19:17 CDT
Reported: 09/25/2020 / 03:58 CDT
CLIENT SERVICES: 866.697.8378
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
medMATCH
= Consistent = Inconsistent
= Prescribed
Test Name
09/24/2020
09/22/2020 09/19/2020 09/17/2020
Marijuana Metabolite POSITIVE POSITIVE NEGATIVE CONFIRMED POSITIVE
Marijuana Metabolite 5
5 NEGATIVE 100
Methadone Metabolite POSITIVE POSITIVE POSITIVE POSITIVE
EDDP 100 NEGATIVE
100 100
Methadone 100
100
NEGATIVE
100
Oxycodone POSITIVE POSITIVE
NEGATIVE
POSITIVE
Noroxycodone 100
100
-
100
Oxycodone 100
100
-
100
Oxymorphone 100
100
-
100
Specimen Validity Testing
Specific Gravity 1.002 L 1.002 L 1.002 L 1.002 L
pH 6.7 6.7 6.7 6.7
Creatinine 11.2 L 12.3 L 14.5 L 11.2 L
Oxidant NEGATIVE NEGATIVE NEGATIVE NEGATIVE
Only results from the current report and the past 4 orders with Quest Diagnostics are displayed (Data includes results meeting strict identification matching). Table displays presumptive
results first then corresponding definitive results. Dash indicates a presumptive test was negative therefore definitive testing was not needed, or the test was not ordered by the clinician
in subsequent testing.
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
LIST OF RESULTS PRINTED IN THE OUT OF RANGE COLUMN:
Creatinine 11.2 L
> or = 20.0 mg/dL IG
Specific Gravity 1.002 L
> or = 1.003 IG
PAGE 3 OF 3
+
+
+
+
+
X
medMATCH
®
legend
If results are ordered with medMATCH
®
, the medMATCH
®
icons will be
displayed next to the appropriate results.
Our results history feature displays results for the current order as well as
results from the prior 4 orders, thus providing additional insights and allowing you to easily
compare current results with historical results and further aid patient care decisions.

Results history
Data includes results meeting strict identification matching.
Patient Information Specimen Information Client Information
Fasting: Y
Specimen:
Collected: 09/24/2020 / 12:00 CDT
Received: 09/24/2020 / 19:17 CDT
Reported: 09/25/2020 / 03:58 CDT
CLIENT SERVICES: 866.697.8378
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
medMATCH
= Consistent = Inconsistent
= Prescribed
Test Name
09/24/2020
09/22/2020 09/19/2020 09/17/2020
Marijuana Metabolite POSITIVE POSITIVE NEGATIVE CONFIRMED POSITIVE
Marijuana Metabolite 5
5 NEGATIVE 100
Methadone Metabolite POSITIVE POSITIVE POSITIVE POSITIVE
EDDP 100 NEGATIVE
100 100
Methadone 100
100
NEGATIVE
100
Oxycodone POSITIVE POSITIVE
NEGATIVE
POSITIVE
Noroxycodone 100
100
-
100
Oxycodone 100
100
-
100
Oxymorphone 100
100
-
100
Specimen Validity Testing
Specific Gravity 1.002 L 1.002 L 1.002 L 1.002 L
pH 6.7 6.7 6.7 6.7
Creatinine 11.2 L 12.3 L 14.5 L 11.2 L
Oxidant NEGATIVE NEGATIVE NEGATIVE NEGATIVE
Only results from the current report and the past 4 orders with Quest Diagnostics are displayed (Data includes results meeting strict identification matching). Table displays presumptive
results first then corresponding definitive results. Dash indicates a presumptive test was negative therefore definitive testing was not needed, or the test was not ordered by the clinician
in subsequent testing.
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
LIST OF RESULTS PRINTED IN THE OUT OF RANGE COLUMN:
Creatinine 11.2 L
> or = 20.0 mg/dL IG
Specific Gravity 1.002 L
> or = 1.003 IG
PAGE 3 OF 3
+
+
+
+
+
X
Table display
• Presumptive results are displayed first, followed by definitive results.
• A dash indicates a presumptive test was negative; therefore, definitive
testing was not performed or the test was not ordered.
Our results history feature displays results for the current order as well as
results from the prior 4 orders, thus providing additional insights and allowing you to easily
compare current results with historical results and further aid patient care decisions.

Results history
Data includes results meeting strict identification matching.
Patient Information Specimen Information Client Information
Fasting: Y
Specimen:
Collected: 09/24/2020 / 12:00 CDT
Received: 09/24/2020 / 19:17 CDT
Reported: 09/25/2020 / 03:58 CDT
CLIENT SERVICES: 866.697.8378
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
medMATCH
= Consistent = Inconsistent
= Prescribed
Test Name
09/24/2020
09/22/2020 09/19/2020 09/17/2020
Marijuana Metabolite POSITIVE POSITIVE NEGATIVE CONFIRMED POSITIVE
Marijuana Metabolite 5
5 NEGATIVE 100
Methadone Metabolite POSITIVE POSITIVE POSITIVE POSITIVE
EDDP 100 NEGATIVE
100 100
Methadone 100
100
NEGATIVE
100
Oxycodone POSITIVE POSITIVE
NEGATIVE
POSITIVE
Noroxycodone 100
100
-
100
Oxycodone 100
100
-
100
Oxymorphone 100
100
-
100
Specimen Validity Testing
Specific Gravity 1.002 L 1.002 L 1.002 L 1.002 L
pH 6.7 6.7 6.7 6.7
Creatinine 11.2 L 12.3 L 14.5 L 11.2 L
Oxidant NEGATIVE NEGATIVE NEGATIVE NEGATIVE
Only results from the current report and the past 4 orders with Quest Diagnostics are displayed (Data includes results meeting strict identification matching). Table displays presumptive
results first then corresponding definitive results. Dash indicates a presumptive test was negative therefore definitive testing was not needed, or the test was not ordered by the clinician
in subsequent testing.
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
LIST OF RESULTS PRINTED IN THE OUT OF RANGE COLUMN:
Creatinine 11.2 L
> or = 20.0 mg/dL IG
Specific Gravity 1.002 L
> or = 1.003 IG
PAGE 3 OF 3
+
+
+
+
+
X
Current results
• The first column includes the current results.
• In this example, the current results were ordered without medMATCH
®
as indicated by lack of icons.
Our results history feature displays results for the current order as well as
results from the prior 4 orders, thus providing additional insights and allowing you to easily
compare current results with historical results and further aid patient care decisions.
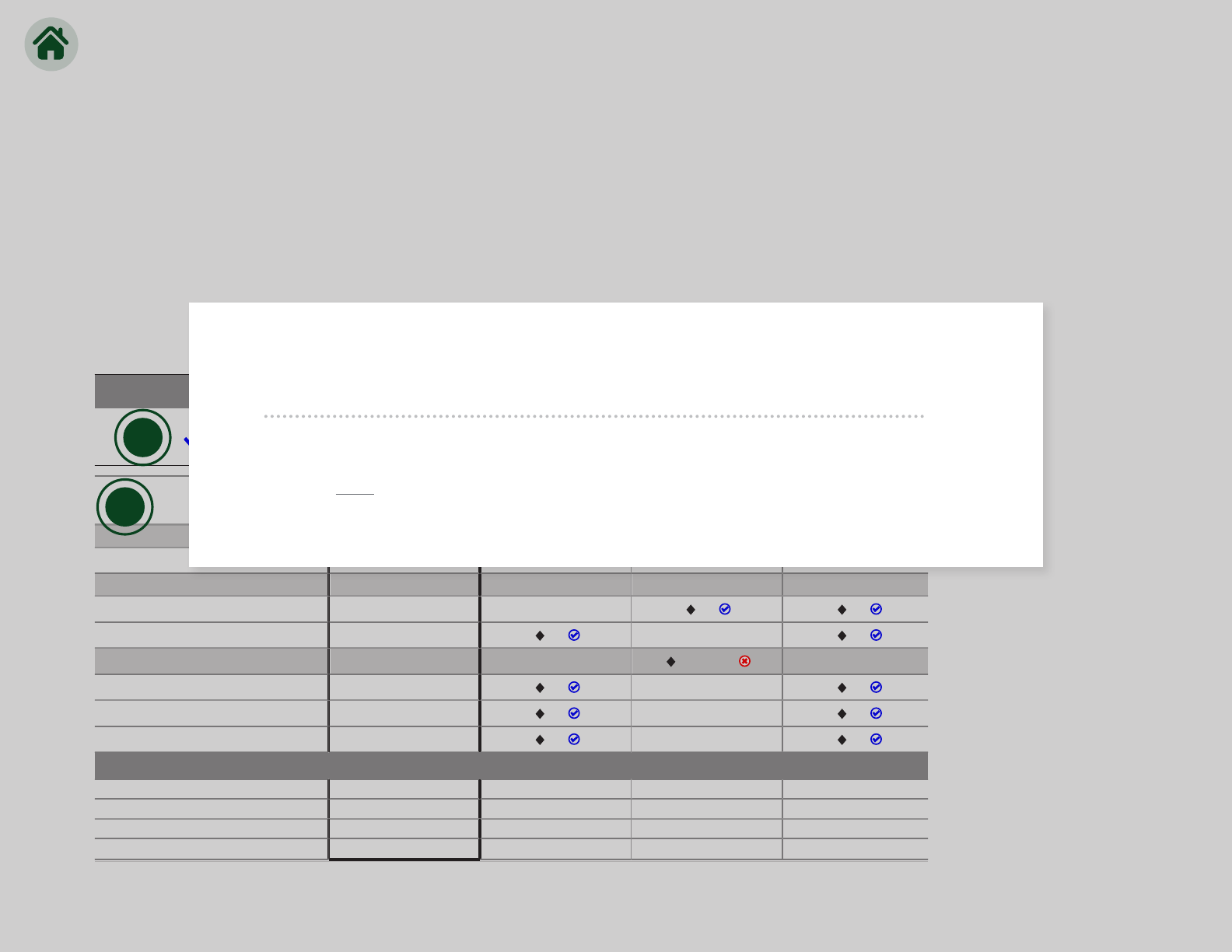
Results history
Data includes results meeting strict identification matching.
Patient Information Specimen Information Client Information
Fasting: Y
Specimen:
Collected: 09/24/2020 / 12:00 CDT
Received: 09/24/2020 / 19:17 CDT
Reported: 09/25/2020 / 03:58 CDT
CLIENT SERVICES: 866.697.8378
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
medMATCH
= Consistent = Inconsistent
= Prescribed
Test Name
09/24/2020
09/22/2020 09/19/2020 09/17/2020
Marijuana Metabolite POSITIVE POSITIVE NEGATIVE CONFIRMED POSITIVE
Marijuana Metabolite 5
5 NEGATIVE 100
Methadone Metabolite POSITIVE POSITIVE POSITIVE POSITIVE
EDDP 100 NEGATIVE
100 100
Methadone 100
100
NEGATIVE
100
Oxycodone POSITIVE POSITIVE
NEGATIVE
POSITIVE
Noroxycodone 100
100
-
100
Oxycodone 100
100
-
100
Oxymorphone 100
100
-
100
Specimen Validity Testing
Specific Gravity 1.002 L 1.002 L 1.002 L 1.002 L
pH 6.7 6.7 6.7 6.7
Creatinine 11.2 L 12.3 L 14.5 L 11.2 L
Oxidant NEGATIVE NEGATIVE NEGATIVE NEGATIVE
Only results from the current report and the past 4 orders with Quest Diagnostics are displayed (Data includes results meeting strict identification matching). Table displays presumptive
results first then corresponding definitive results. Dash indicates a presumptive test was negative therefore definitive testing was not needed, or the test was not ordered by the clinician
in subsequent testing.
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
LIST OF RESULTS PRINTED IN THE OUT OF RANGE COLUMN:
Creatinine 11.2 L
> or = 20.0 mg/dL IG
Specific Gravity 1.002 L
> or = 1.003 IG
PAGE 3 OF 3
+
+
+
+
+
X
Previous results
Up to 4 prior results are displayed. In this example, prior results were
ordered with medMATCH
®
as indicated by inclusion of icons.
Our results history feature displays results for the current order as well as
results from the prior 4 orders, thus providing additional insights and allowing you to easily
compare current results with historical results and further aid patient care decisions.

Result details
Drug Monitoring Report
Red test name and result indicate a positive test result.
Report Status: Final
REPORT3
Patient Information Specimen Information Client Information
SAMPLE REPORT3
DOB: Not Given AGE: 40
Gender: M Fasting: Y
Phone: NG
Patient ID: 0131634N
Specimen: IF048807B
Requisition: 0131634
Lab Ref #: IF048807B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 13:10 EDT
Reported: 09/29/2020 / 13:19 EDT
Client #: 97502882
TEST
Attn: Dr. Jones
Main Street
City, NJ 07000
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048807B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Drug Monitoring Report
Test Ordered Result Cutoff Lab
DRUG MONITOR,AMPHETAMINE, W/CONF, W/DL,URINE IG
Amphetamines POSITIVE 500 ng/mL
Amphetamine 300 250 ng/mL
Methamphetamine 500 250 ng/mL
Amphetamines Comments: See Amphetamines Notes, LDT Notes
DRUG MONITOR, METHAMPHETAMINE D/L,U IG
d Methamphetamine 80 %
l Methamphetamine 20 %
D/L Methamphet Comments: See D/L Methamphet Notes, LDT Notes
NOTES AND COMMENTS
This drug testing is for medical treatment only. Analysis was performed as non-forensic testing and these results should be used only by healthcare providers to
render diagnosis or treatment, or to monitor progress of medical conditions.
Amphetamines Notes:
Amphetamine detected is consistent with the use of the drug Amphetamine. Amphetamine can be a prescribed drug and is also a metabolite of methamphetamine.
Amphetamine, Methamphetamine detected is consistent with the use of the drug Methamphetamine.
D/L Methamphet Notes:
d-Methamphetamine > 20% detected is consistent with the use of the drug(s) prescription Methamphetamine, Benzphetamine or Methamphetamine from a
clandestine source.
LDT Notes:
Confirmation tests were developed and their analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or
approved by the FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
Healthcare Providers needing Interpretation assistance, please contact us at 1.877.40.RXTOX (1.877.407.9869) M-F, 8am to 10pm EST
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
PAGE 1 OF 1
+
+
When results are ordered without medMATCH, the Drug Monitoring report will be issued.
The body of the Drug Monitoring report provides the individual test results, with key
results identified with icons and color to aid in fast and intuitive result interpretation.
+
X
Drug Monitoring Report
Enhancements have also been applied to non-medMATCH
®
test orders.

Result details
Drug Monitoring Report
Red test name and result indicate a positive test result.
Report Status: Final
REPORT3
Patient Information Specimen Information Client Information
SAMPLE REPORT3
DOB: Not Given AGE: 40
Gender: M Fasting: Y
Phone: NG
Patient ID: 0131634N
Specimen: IF048807B
Requisition: 0131634
Lab Ref #: IF048807B
Collected: 09/29/2020 / 11:00 EDT
Received: 09/29/2020 / 13:10 EDT
Reported: 09/29/2020 / 13:19 EDT
Client #: 97502882
TEST
Attn: Dr. Jones
Main Street
City, NJ 07000
CLIENT SERVICES: 866.697.8378 SPECIMEN: IF048807B
Quest, Quest Diagnostics, the associated logo and all associated Quest Diagnostics marks are the trademarks of Quest Diagnostics.
Drug Monitoring Report
Test Ordered Result Cutoff Lab
DRUG MONITOR,AMPHETAMINE, W/CONF, W/DL,URINE IG
Amphetamines POSITIVE 500 ng/mL
Amphetamine 300 250 ng/mL
Methamphetamine 500 250 ng/mL
Amphetamines Comments: See Amphetamines Notes, LDT Notes
DRUG MONITOR, METHAMPHETAMINE D/L,U IG
d Methamphetamine 80 %
l Methamphetamine 20 %
D/L Methamphet Comments: See D/L Methamphet Notes, LDT Notes
NOTES AND COMMENTS
This drug testing is for medical treatment only. Analysis was performed as non-forensic testing and these results should be used only by healthcare providers to
render diagnosis or treatment, or to monitor progress of medical conditions.
Amphetamines Notes:
Amphetamine detected is consistent with the use of the drug Amphetamine. Amphetamine can be a prescribed drug and is also a metabolite of methamphetamine.
Amphetamine, Methamphetamine detected is consistent with the use of the drug Methamphetamine.
D/L Methamphet Notes:
d-Methamphetamine > 20% detected is consistent with the use of the drug(s) prescription Methamphetamine, Benzphetamine or Methamphetamine from a
clandestine source.
LDT Notes:
Confirmation tests were developed and their analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or
approved by the FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
Healthcare Providers needing Interpretation assistance, please contact us at 1.877.40.RXTOX (1.877.407.9869) M-F, 8am to 10pm EST
PERFORMING SITE:
IG QUEST DIAGNOSTICS-IRVING, 4770 REGENT BLVD., IRVING, TX 75063-2445 Laboratory Director: ROBERT L BRECKENRIDGE,MD, CLIA: 45D0697943
PAGE 1 OF 1
+
+
When results are ordered without medMATCH, the Drug Monitoring report will be issued.
The body of the Drug Monitoring report provides the individual test results, with key
results identified with icons and color to aid in fast and intuitive result interpretation.
+
+
X
Drug Class comments and LDT notes
Drug Class Specific Notes and LDT Notes are consolidated and displayed at
the end of the report.
